The Problem. |
More about Polyethylene Terephthalate
We have learnt that PET is a very common thermoplastic polymer in the bottling industry through a visit to a local water bottling company.
It is used for the following reasons:
- Lightweight
- Strong
- Easy to produce
- Recyclable
- Cheap
Problem #1 - Degradation of PET
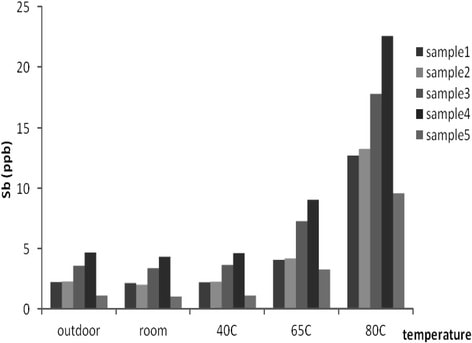
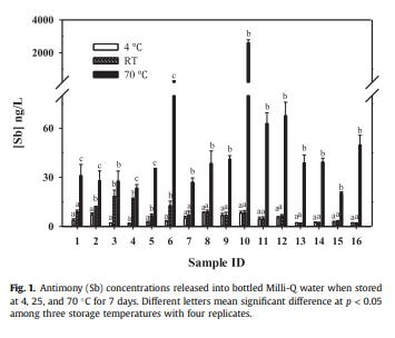
Through professional research, the chemical content of bottled water varies when the bottles are stored in different storage conditions. Experiments have shown that the Antimony (Sb), Bisphenol A and Acetaldehyde levels rise in correlation to the temperature of which the bottle is exposed to.
Experimental data has shown the increase of Antimony (Sb) concentrations when the bottle is exposed to higher temperatures
FINDING: Antimony concentrations in water increase as the temperature of the bottle increases.
Problem #2 - Handling of bottled water
There are three stages a plastic bottle goes through.
Stage 1 - Bottling Plant
|
|
During our research phase, we have visited Bio Osmo Berhad, a bottled water company here in Malaysia to learn about the bottling water process.
The Ministry of Health Malaysia (MOH) has enforced some very stringent health and safety guidelines compliant to international standards. The guidelines include that the bottled water and contents must not be exposed to direct sunlight and has to be kept in suitable dry conditions. Therefore, the bottled water is safe to consume when it is in the bottling plant. |
Stage 2 - Transport
|
The problem begins when the bottled water leaves the plant into transport phase. This is because there are no longer any guidelines regarding the handling of the bottled water. As a result, they are stored in unsuitable conditions.
Through surveys, the internal temperature of a truck has the potential to rise to up to 80 degrees Celsius with a ambient temperature of 35 degrees Celsius. This means the average Antimony concentration will exceed the Maximum Acceptable Limit in just a few hours. |
Stage 3 - Retail and Consumer
|
Bottled water is sold to consumers at places such as convenience stores and petrol stations. And through personal experiences of us and survey participants, there are witness to the bottled water stored outdoors under direct sunlight.
Our survey response showed majority having consumed bottled water which is stored in cars. |
What is Antimony?
Antimony is a heavy metal. Intake of Antimony should be as low as possible because it is a substance considered as potentially carcinogenic by the International Agency for Research on Cancer (IARC) as well as the National Cancer Institute. In addition, Antimony intake can cause respiratory irritation, pneumoconiosis, gastrointestinal symptoms and cardiac arrhythmias.
Conclusion
|
There are very significant effects of the storage temperature of the bottles on the Antimony (Sb) concentrations present in the water.
|
Bottled water might not be handled appropriately after leaving the bottling plant. It could be exposed to extremely high temperatures. Causing higher amounts of Antimony to be released.
|